
SOLVED: (b) Concentration of nitrite = 1.74 ppm * 69.027 / 14.007 = 8.574 ppm. This can be transformed into molar concentration as 8.574 μg/mL * (1 g / 1 μg) * (1 mL / 69.027 g) * (1 mol / 12 * 10^2 mL) = 8.574 ppm * (1 / 1 * 10^24) mol.
For "16.8 V" H2O2 solution ( density = 0.95 g/ml), calculate : 1. Molarity 2. Molality 3. 4. 5. ppm of solute 6. mole fraction of solute

Solution Concentration Chemistry. Ways to Express Solution Composition Molarity: Mass %: Volume %: Parts per million (ppm): - ppt download
For "16.8 V" H2O2 solution ( density = 0.95 g/ml), calculate : 1. Molarity 2. Molality 3. 4. 5. ppm of solute 6. mole fraction of solute
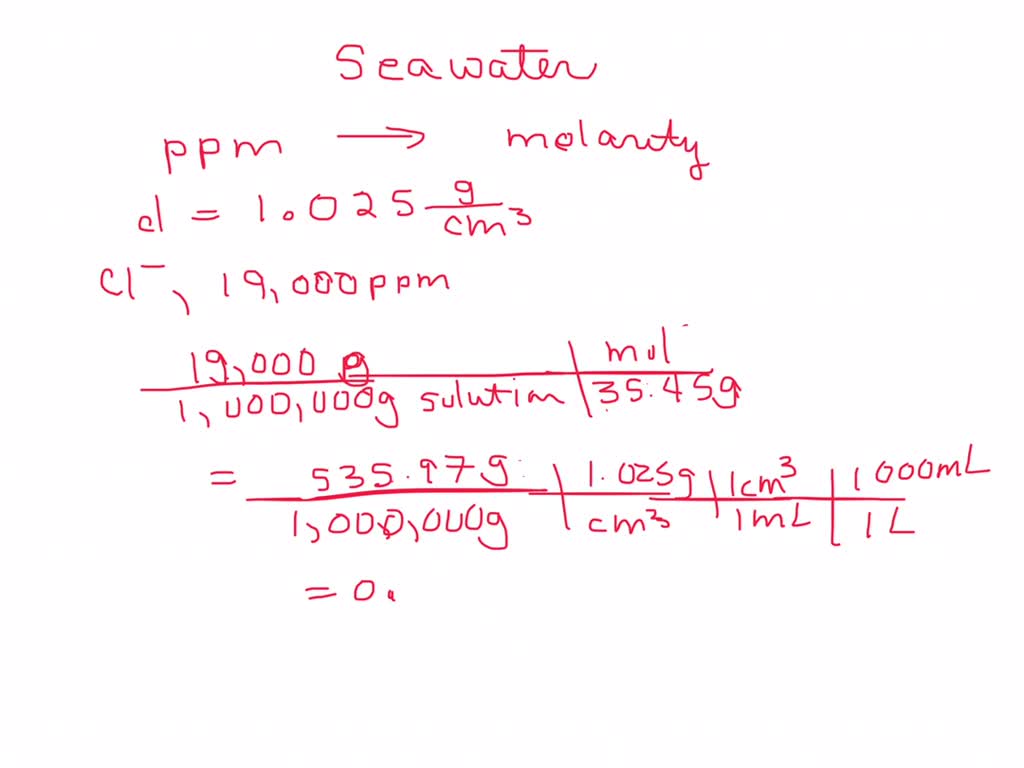
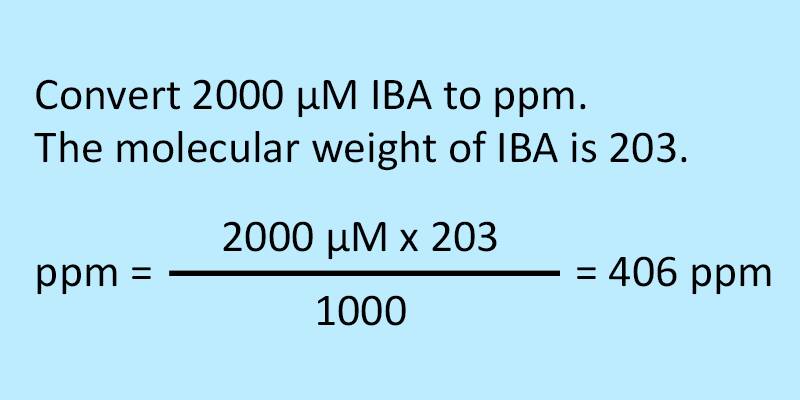
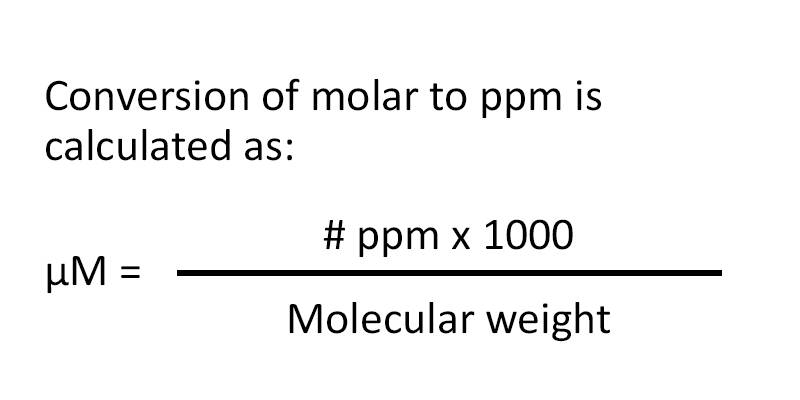






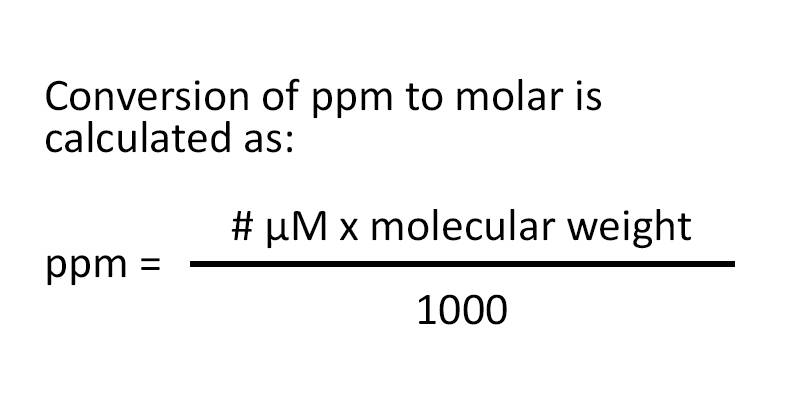







![Telugu] Hardness of water is 200 ppm. Calculate the molarity and norm Telugu] Hardness of water is 200 ppm. Calculate the molarity and norm](https://static.doubtnut.com/ss/web-overlay-thumb/2619800.webp)



